8th Grade > Physics
CHEMICAL EFFECTS OF ELECTRIC CURRENT MCQs
:
C
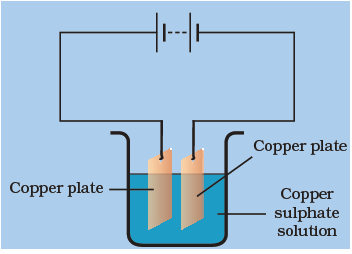
When electric current is passed through the copper sulphate solution, copper sulphate dissociates into Cu2+ and SO2−4 ions. The Cu2+ get drawn to the electrode connected to the negative terminal of the battery and gets deposited on it.
:
B
A solution can conduct electricity when it has ions which act as charge carriers. When sugar is added to water, it does not produce ions to facilitate electrical conductivity. On the other hand, a salt solution has ions to conduct electricity. So, a sugar solution is a poor conductor as compared to a salt solution.
:
A
A material is said to be a good conductor if it allows electricity to pass through it easily. Materials which are classified as poor conductors does not also allow electricity to pass through them easily. However, if the electrical source is strong enough, it may allow to pass electric current through it. For example, air is a poor conductor of electricity. However, during lightning, air allows the electric current to pass through it.
:
C
Pure water that is completely free of all salts is a bad conductor of electricity. Distilled water is an example. To make it conducting, it should have ions to carry charges. Substances like salts, acids and bases when dissolved in water dissociate to form ions. So, they conduct electricity.
:
C
When the circuit is closed, charges start flowing from one end to the other. While checking the LED, the circuit should not be closed for a long time. Otherwise, the cells of the battery will drain very quickly as this offers very little resistance to the flow of current.
:
C
The electric current in a metal wire is nothing but the flow of electrons. When these electrons pass through the filament of the bulb, it produces heat and thereby light energy. Hence, this heating effect of electric current is responsible for the glow.
:
A
When two or more cells are connected together end-to-end, it makes a battery. The typical advantage of connecting cells in a battery is to add the voltage of all the cells together to get a higher voltage.
:
B
Rusting of iron takes place in the presence of oxygen, moisture present in the air. A coating of zinc is deposited on the iron to protect it from corrosion and formation of rust. The layer of zinc prevents oxygen from reacting with the iron, making the iron object last much longer.
:
C
LED stands for Light Emitting Diode. Compared to an electric bulb, it requires less current for it to glow. It is used in a tester to check the conductivity of the liquid.
:
A
Pure water that is completely free of all salts is a bad conductor of electricity. To make it conducting, it should have ions to carry charges. Salts, when dissolved in water, can produce ions that can carry electricity. The chemical formula of common salt is NaCl (Sodium Chloride). It dissociates into sodium ion and chloride ion which act like charge carriers to conduct electricity.
Sugar, glucose, and kerosene do not form ions in water and cannot conduct electricity.