12th Grade > Physics
HEAT TRANSFER MCQs
Total Questions : 30
| Page 2 of 3 pages
Answer: Option D. -> 1800
:
D
The energy radiated per second by a black body is given by Stefan's Law as Et=σT2×A where A is the surface area of the black body
EtσT4×4πr2
Since black body is a sphere, (A=4πr2).
Case(i)
Et=450,T=500K,r=0.12m,
∴450=4πσ(500)4(0.12)2 .....(i)
Case(ii)
Et=?,T=1000K,r=0.06m,
dividing (ii) and (I) we get,
Et450=(1000)4(0.06)2(500)4(0.12)2=2422=4
⇒Et=450×4=1800W
(d) is the correct option.
:
D
The energy radiated per second by a black body is given by Stefan's Law as Et=σT2×A where A is the surface area of the black body
EtσT4×4πr2
Since black body is a sphere, (A=4πr2).
Case(i)
Et=450,T=500K,r=0.12m,
∴450=4πσ(500)4(0.12)2 .....(i)
Case(ii)
Et=?,T=1000K,r=0.06m,
dividing (ii) and (I) we get,
Et450=(1000)4(0.06)2(500)4(0.12)2=2422=4
⇒Et=450×4=1800W
(d) is the correct option.
Answer: Option B. -> 6.1 min
:
B
As the temperature differences are small, we can use Newton's law of cooling.
dθdt=−kt(θ−θ0)
dθθ−θ0=−kdt
or, ...(i)
Where k is a constant, θ is the temperature of the body at time t and θ0=16∘C is the temperature of the surrounding. We have,
∫36∘C40∘Cdθθ−θ0=−kt(5min)
ln36∘C−16∘C40∘C−16∘C=−kt(5min)
or,
k=−ln(56)5min
or, .
If t be the time required for the temperature to fall from 36∘C to 32∘C then by (i),
∫32∘C36∘Cdθθ−θ0=−kt
or, ln32∘C−16∘C36∘C−16∘C=−ln(56)5min
or, t=ln(45)(ln(56)×5min
= 6.1 min.
Alternative method
The mean temperature of the body as it cools from 40∘C to 36∘C is 40∘C+36∘C2=38∘C The rate of decrease of temperature is
40∘C+36∘C5min=0.80∘C/min .
Newton's law of cooling is
dθdt=−k(θ−θ0)
or, -0.8∘C/min = - k(38∘C - 16∘C)=-k(22∘C)
or, k=0.822min−1.
Let the time taken for the temperature to become 32∘C be t.
During this period,.
The mean temperature is
Now,
:
B
As the temperature differences are small, we can use Newton's law of cooling.
dθdt=−kt(θ−θ0)
dθθ−θ0=−kdt
or, ...(i)
Where k is a constant, θ is the temperature of the body at time t and θ0=16∘C is the temperature of the surrounding. We have,
∫36∘C40∘Cdθθ−θ0=−kt(5min)
ln36∘C−16∘C40∘C−16∘C=−kt(5min)
or,
k=−ln(56)5min
or, .
If t be the time required for the temperature to fall from 36∘C to 32∘C then by (i),
∫32∘C36∘Cdθθ−θ0=−kt
or, ln32∘C−16∘C36∘C−16∘C=−ln(56)5min
or, t=ln(45)(ln(56)×5min
= 6.1 min.
Alternative method
The mean temperature of the body as it cools from 40∘C to 36∘C is 40∘C+36∘C2=38∘C The rate of decrease of temperature is
40∘C+36∘C5min=0.80∘C/min .
Newton's law of cooling is
dθdt=−k(θ−θ0)
or, -0.8∘C/min = - k(38∘C - 16∘C)=-k(22∘C)
or, k=0.822min−1.
Let the time taken for the temperature to become 32∘C be t.
During this period,.
The mean temperature is
Now,
Answer: Option C. -> 955 K
:
C
Let the temperature of the coil be T. The coil will emit radiation at a rate AσT4. Thus,
1000W =(0.020m2)×(6.0×12−8W/m2−K4)×T64
or, T4=10000.020×6.00×10−8K4
=8.33×1011K4
or, T=955K.
:
C
Let the temperature of the coil be T. The coil will emit radiation at a rate AσT4. Thus,
1000W =(0.020m2)×(6.0×12−8W/m2−K4)×T64
or, T4=10000.020×6.00×10−8K4
=8.33×1011K4
or, T=955K.
Question 14. A copper sphere is suspended in an evacuated chamber maintained at 300 K. The sphere is maintained at a constant temperature of 500 K by heating it electrically. A total of 210 W of electric power is needed to do it. When the surface of the copper sphere is completely blackened, 700 W is needed to maintain the same temperature of the sphere. Calculate the emissivity of copper.
Answer: Option C. -> 0.3
:
C
Before painting it black the power needed = eσA(T4−T40)=210 ...(i)
T = 500 K
T0 = 300 K
e → emissivity
After the paint the surface behaves like a black body.
∴σA(T4−T40)=700 ...(ii)
Dividing (i) and (ii)
e=210700
⇒ e = 0.3
:
C
Before painting it black the power needed = eσA(T4−T40)=210 ...(i)
T = 500 K
T0 = 300 K
e → emissivity
After the paint the surface behaves like a black body.
∴σA(T4−T40)=700 ...(ii)
Dividing (i) and (ii)
e=210700
⇒ e = 0.3
Answer: Option B. -> 887 J
:
B
Stefan's law: ΔQΔt=eσAT4
For an ideal radiator e = 1 ⇒ΔQΔt=6×10−8×1.6×(273+37)4
=6×10−8×1.6×314×104
= 887 J per second.
:
B
Stefan's law: ΔQΔt=eσAT4
For an ideal radiator e = 1 ⇒ΔQΔt=6×10−8×1.6×(273+37)4
=6×10−8×1.6×314×104
= 887 J per second.
Answer: Option A. -> 25681
:
A
According to Wien's law wavelength corresponding to maximum energy decreases. When the temperature of blackbody increases i.e.,
λmT=constant⇒T2T1=λ1λ2=λ03λ04=43
Now according to Stefan's law
E2E1=(T2T1)4=(43)4=25681.
:
A
According to Wien's law wavelength corresponding to maximum energy decreases. When the temperature of blackbody increases i.e.,
λmT=constant⇒T2T1=λ1λ2=λ03λ04=43
Now according to Stefan's law
E2E1=(T2T1)4=(43)4=25681.
Answer: Option C. -> Spherical
:
C
For all modes of heat transfer ΔQ∝Area,A.So we should choose the shape with minimum area for minimum heat transfer.
(a)For cubical:
Volume, V = a3=1l,∴a=1dm(decameter)
Area, A = 6a2=6dm2
(b)For conical: V= πr2h3, but given r=h,r=313π dm
A=πr(√2r)+πr2
= 7.354 dm2
(c)For Sphere:
V=43πr3, ∴r=(34)13dm
A=4πr2=4.84dm2
(d) For Cylinder:
V=π×r2×r∴r=1π13dm
A=2πr2+2πr.r=5.86dm2
As Sphere has the least area among these, it will be the shape with minimum heat loss. In fact sphere is the shape with the minimum surface area among all possible shapes!
:
C
For all modes of heat transfer ΔQ∝Area,A.So we should choose the shape with minimum area for minimum heat transfer.
(a)For cubical:
Volume, V = a3=1l,∴a=1dm(decameter)
Area, A = 6a2=6dm2
(b)For conical: V= πr2h3, but given r=h,r=313π dm
A=πr(√2r)+πr2
= 7.354 dm2
(c)For Sphere:
V=43πr3, ∴r=(34)13dm
A=4πr2=4.84dm2
(d) For Cylinder:
V=π×r2×r∴r=1π13dm
A=2πr2+2πr.r=5.86dm2
As Sphere has the least area among these, it will be the shape with minimum heat loss. In fact sphere is the shape with the minimum surface area among all possible shapes!
Answer: Option C. -> 19.1 hours
:
C
t=QlKA(θ1−θ2)=mLlKA(θ1−θ2)=VρLlKA(θ1−θ2)
=5×A×0.92×80×5+1020.004×A×10×3600=19.1hours.
:
C
t=QlKA(θ1−θ2)=mLlKA(θ1−θ2)=VρLlKA(θ1−θ2)
=5×A×0.92×80×5+1020.004×A×10×3600=19.1hours.
Answer: Option B. -> A and B will represent cooling curves of water and oil respectively
:
B
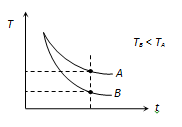
As we know, Rate of cooling ∝1specificheat(c)
∵Coil<Cwater
⇒(Rateofcooling)oil>(Rateofcooling)water
It is clear that, at a particular time after start cooling, temperature of oil will be less than that of water.
So graph B represents the cooling curve of oil and A represents the cooling curve of water.
:
B

As we know, Rate of cooling ∝1specificheat(c)
∵Coil<Cwater
⇒(Rateofcooling)oil>(Rateofcooling)water
It is clear that, at a particular time after start cooling, temperature of oil will be less than that of water.
So graph B represents the cooling curve of oil and A represents the cooling curve of water.
Question 20. One end of a copper rod of length 1.0 m and area of cross-section 10−3 is immersed in boiling water and the other end in ice. If the coefficient of thermal conductivity of copper is 92cal/m−s−∘C and the latent heat of ice is 8×104cal/kg, then the amount of ice which will melt in one minute is
Answer: Option C. -> 6.9×10−3kg
:
C
Heat transferred in one minute is utilised in melting the ice so, KA(θ1−θ2)tl=m×L
⇒m=10−3×92×(100−0)×601×8×104=6.9×10−3kg
:
C
Heat transferred in one minute is utilised in melting the ice so, KA(θ1−θ2)tl=m×L
⇒m=10−3×92×(100−0)×601×8×104=6.9×10−3kg

















