Question
Water and turpentine oil (specific heat less than that of water) are both heated to same temperature. Equal amounts of these placed in identical calorimeters are then left in air


Answer: Option B
:
B
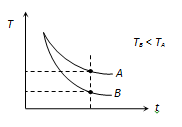
As we know, Rate of cooling ∝1specificheat(c)
∵Coil<Cwater
⇒(Rateofcooling)oil>(Rateofcooling)water
It is clear that, at a particular time after start cooling, temperature of oil will be less than that of water.
So graph B represents the cooling curve of oil and A represents the cooling curve of water.
Was this answer helpful ?
:
B

As we know, Rate of cooling ∝1specificheat(c)
∵Coil<Cwater
⇒(Rateofcooling)oil>(Rateofcooling)water
It is clear that, at a particular time after start cooling, temperature of oil will be less than that of water.
So graph B represents the cooling curve of oil and A represents the cooling curve of water.
Was this answer helpful ?


















Submit Solution