10th Grade > Chemistry
METALS AND NON-METALS MCQs
Total Questions : 57
| Page 2 of 6 pages
Answer: Option C. -> Graphite
:
C
Graphite is a good conductor of electricity because of itsstructure. Like metals, it containsdelocalisedelectrons which are free to conduct electricity.
:
C
Graphite is a good conductor of electricity because of itsstructure. Like metals, it containsdelocalisedelectrons which are free to conduct electricity.
Answer: Option B. -> silver sulphide
:
B
Corrosion is a process of conversion of a metal into its compound ie. oxides or carbonates etc.by continuous interaction with atmospheric air and water vapour.
On exposure to air for a long time, silver articles combine with sulphur in the atmosphere. As a result, black coating of silver sulphide is formed on their surface. The reaction involved is:
2Ag(s)+H2S(g)→Ag2S(s)+H2(g)
:
B
Corrosion is a process of conversion of a metal into its compound ie. oxides or carbonates etc.by continuous interaction with atmospheric air and water vapour.
On exposure to air for a long time, silver articles combine with sulphur in the atmosphere. As a result, black coating of silver sulphide is formed on their surface. The reaction involved is:
2Ag(s)+H2S(g)→Ag2S(s)+H2(g)
Answer: Option A. -> Fe
:
A
Fe (Iron) is the main constituent of haemoglobin. It helps in carrying oxygen to the cells.
:
A
Fe (Iron) is the main constituent of haemoglobin. It helps in carrying oxygen to the cells.
Answer: Option A. -> True
:
A
Magnalium is analloy of aluminium with magnesium and small amounts of nickel and tin.
:
A
Magnalium is analloy of aluminium with magnesium and small amounts of nickel and tin.
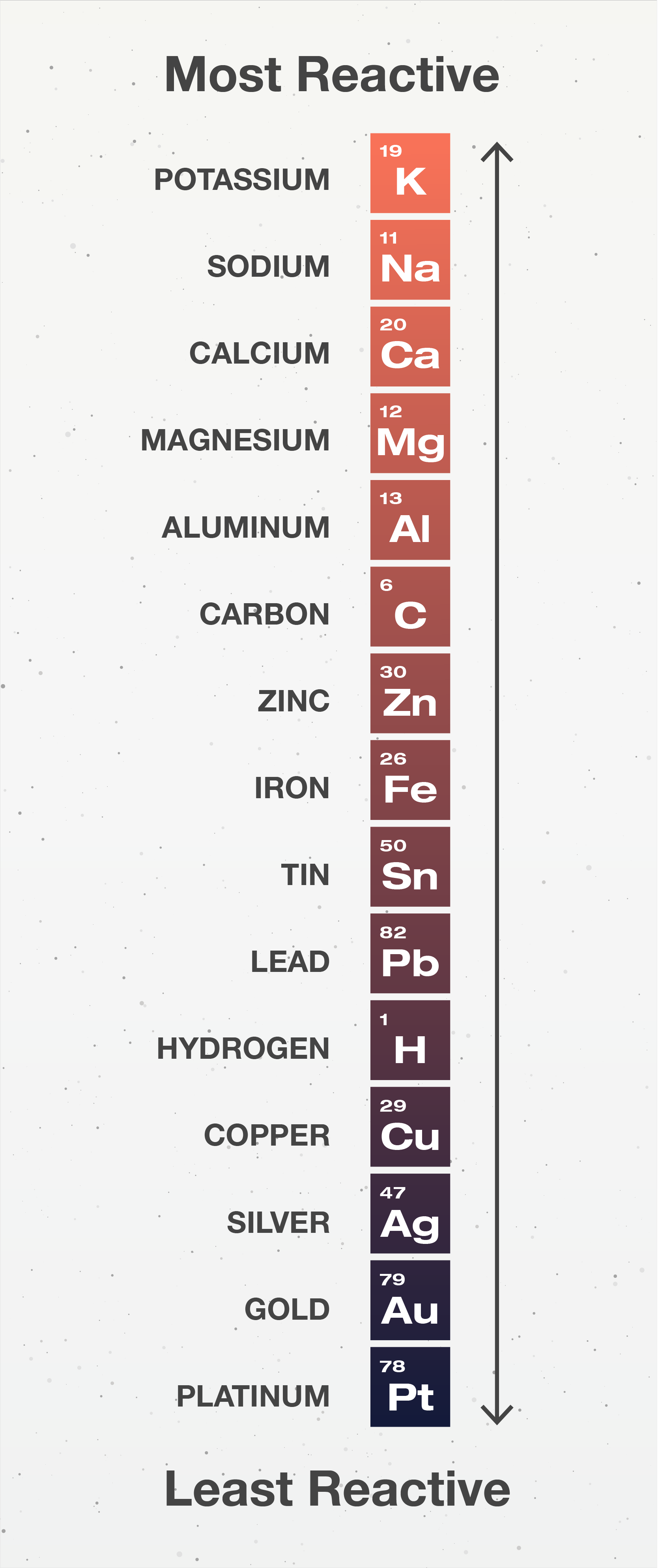
Answer: Option C. -> Metals above hydrogen in the reactivity series cannot displace hydrogen from dilute acids.
:
C
For example: Fe(s)+CuSO4(aq)→FeSO4(aq)+Cu(s)
Here, ironis more reactive than copper. So, itdisplaces copperfrom its solution.
:
C
- Alloys that contain mercury are known as amalgams.
- The metals high up in the reactivity series are very reactive.
- More reactive metals can replace less reactive metals.
- Metals which are above hydrogen in the reactivity series will displace hydrogen from diluteacids.
For example: Fe(s)+CuSO4(aq)→FeSO4(aq)+Cu(s)
Here, ironis more reactive than copper. So, itdisplaces copperfrom its solution.
Answer: Option A. -> True
:
A
Mercury is the only metal that exists as a liquid at room temperature and pressure.
:
A
Mercury is the only metal that exists as a liquid at room temperature and pressure.
:
When zinc oxide is heated with carbon, it is reduced to metallic zinc. Carbon acts as the reducing agent.
Answer: Option C. -> Au
:
C
Malleability refers to the ability of a metal to get beaten or flattened into thin sheets. Gold is the most malleable metal of all. Hence, extremely thin sheets of it can be obtained.
This makes it useful in many applications like shielding from harmful radiation, in jewellery, etc.
:
C
Malleability refers to the ability of a metal to get beaten or flattened into thin sheets. Gold is the most malleable metal of all. Hence, extremely thin sheets of it can be obtained.
This makes it useful in many applications like shielding from harmful radiation, in jewellery, etc.
Answer: Option A. -> gangue
:
A
Impurities found in ores mined from the earth are called gangue. It usually consists of large amounts of soil, sand, stones etc.
:
A
Impurities found in ores mined from the earth are called gangue. It usually consists of large amounts of soil, sand, stones etc.