12th Grade > Chemistry
CHEMISTRY IN EVERYDAY LIFE MCQs
Total Questions : 30
| Page 2 of 3 pages
Answer: Option A. -> Both A and R are true and R is a correct explanation of A
:
A
Hard water contains calcium and magnesium ions which forms insoluble precipitate with soap which are sticky in nature
2C17H35COONaSoap+CaCl2→+2NaCl+(C17H35COO)2CaIsoluablepercipitate
:
A
Hard water contains calcium and magnesium ions which forms insoluble precipitate with soap which are sticky in nature
2C17H35COONaSoap+CaCl2→+2NaCl+(C17H35COO)2CaIsoluablepercipitate
Answer: Option B. -> Prevent rapid drying
:
B
Glycerol is added mainly to shaving soaps to prevent rapid drying.
:
B
Glycerol is added mainly to shaving soaps to prevent rapid drying.
Answer: Option B. -> Both A and R are true but R is not a correct explanation of A
:
B
Aspartame is 100 times sweeter than cane sugar. Aspartameis not stable at normal cooking temperature and that’s why it is not used in normal cooking.
:
B
Aspartame is 100 times sweeter than cane sugar. Aspartameis not stable at normal cooking temperature and that’s why it is not used in normal cooking.
Answer: Option C. -> It contains both estrogen and progesterone derivatives
:
C
Birth control pills essentially contain a mixture of synthetic estrogen and progesterone derivatives.
:
C
Birth control pills essentially contain a mixture of synthetic estrogen and progesterone derivatives.
Answer: Option D. -> Alitame
:
D
SweetenerAspartameSaccharinSucraloseAlitameSweetness valuecompared to cane sugar1005506002000
:
D
SweetenerAspartameSaccharinSucraloseAlitameSweetness valuecompared to cane sugar1005506002000
Answer: Option D. -> CH3(CH2)16COO(CH2CH2O)nCH2CH2OH
:
D
Liquid dishwashing detergents are non-ionic type detergents.
Option (a) is soap.
Option (b) is anionic type detergent.
Option (c) is cationic type detergent.
:
D
Liquid dishwashing detergents are non-ionic type detergents.
Option (a) is soap.
Option (b) is anionic type detergent.
Option (c) is cationic type detergent.
Answer: Option B. -> Sucrose
:
B
Except Sucrose, all are artificial sweetener and provide negligible calories. Also these sweeteners are multiple times sweeter than cane sugar. So, they are used in very small quantity.
:
B
Except Sucrose, all are artificial sweetener and provide negligible calories. Also these sweeteners are multiple times sweeter than cane sugar. So, they are used in very small quantity.
Answer: Option B. -> antibiotic broad spectrum
:
B
Chloramphenicol, isolated in 1947, is a broad spectrum antibiotic. It is rapidly absorbed from the gastrointestinal tract and hence can be given orally in case of typhoid, dysentery, acute fever, certain form of urinary infections, meningitis and pneumonia.
:
B
Chloramphenicol, isolated in 1947, is a broad spectrum antibiotic. It is rapidly absorbed from the gastrointestinal tract and hence can be given orally in case of typhoid, dysentery, acute fever, certain form of urinary infections, meningitis and pneumonia.
Question 19. Read the assertion and reason carefully to mark the correct option out of the options given below:
(a)If both assertion and reason are true and the reason is the correct explanation of the assertion.
(b)If both assertion and reason are true but reason is not the correct explanation of the assertion.
(c)If assertion is true but reason is false.
(d)If assertion is false but reason is true.
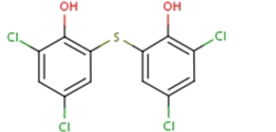
Assertion : Bithional is added to soap for its beautification and solidification.
Reason : Bithional is a sulpha drug.
(a)If both assertion and reason are true and the reason is the correct explanation of the assertion.
(b)If both assertion and reason are true but reason is not the correct explanation of the assertion.
(c)If assertion is true but reason is false.
(d)If assertion is false but reason is true.
Assertion : Bithional is added to soap for its beautification and solidification.
Reason : Bithional is a sulpha drug.
Answer: Option C. -> Non-ionic detergents
:
C
Non-ionic detergents do not contain any ion in their constitution. One such detergent is formed when stearic acid reacts with polyethyleneglycol.
:
C
Non-ionic detergents do not contain any ion in their constitution. One such detergent is formed when stearic acid reacts with polyethyleneglycol.