11th And 12th > Chemistry
CHEMICAL BONDING MCQs
Total Questions : 30
| Page 1 of 3 pages
Answer: Option B. ->
The extent of H-bonding decreases from water to methanol while it is absent in ether
:
B
:
B
Water has strongest hydrogen bond of the three
Answer: Option B. ->
H2O> HF> NH3> CH4
:
B
H2O has higher b.pt among hydrogen bonded molecules
:
B
H2O has higher b.pt among hydrogen bonded molecules
Answer: Option D. ->
A is false but R is true
:
D
Due to stronger hydrogen bonding in water, it has a higher boilind point.
:
D
Due to stronger hydrogen bonding in water, it has a higher boilind point.
Answer: Option B. ->
CH3F, CH4
:
B
:
B
F is not linked to directly to hydrogen
Answer: Option C. ->
I−>Br−>Cl−>F−
:
C
:
C
The polarisability of an anion also depends on its size and charge – the larger the negative ion and the larger its charge the more polarisable it becomes, that is, more is the size of anion more will be the polarisability.
Answer: Option A. ->
NH3> PH3> AsH3> SbH3
:
A
NH3 has sp3 hybridisation with bond angle 1070 where as PH3, AsH3, SbH3 has pure orbitals overlapping with bond angle around 900
:
A
NH3 has sp3 hybridisation with bond angle 1070 where as PH3, AsH3, SbH3 has pure orbitals overlapping with bond angle around 900
Answer: Option D. ->
II > I = III > IV
:
D
:
D
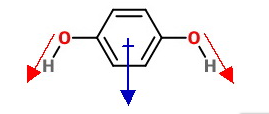
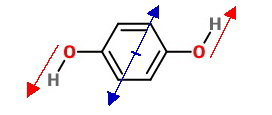
Para compound has zero dipole moment and ortho compound has maximum value.
If we calculate the vector addition, magnitudes of dipole moment for I and III turn
out to be the same.
Answer: Option B. ->
BeCl2> SO2
:
B
BeCl2 is linear with zero dipole moment & SO2 is angular with dipole moment
:
B
BeCl2 is linear with zero dipole moment & SO2 is angular with dipole moment
Answer: Option D. ->
Increases progressively
:
D
From 109.28 to 120 to 180 degrees.
:
D
From 109.28 to 120 to 180 degrees.