12th Grade > Chemistry
ALCOHOLS PHENOLS AND ETHERS MCQs
Total Questions : 28
| Page 1 of 3 pages
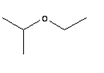
Answer: Option C. -> 2-Ethoxy propane
:
C
Its an ether. Longest chain is propane. Ethers are named as derivatives of alkane.
:
C
Its an ether. Longest chain is propane. Ethers are named as derivatives of alkane.
Answer: Option D. -> methyl iodide & phenol
:
D
In case of anisole, methylphenyl oxonium ion, is formed by protonation of ether. The bond between O–CH3 is weaker than the bond betweenO–C6H5 because the carbon of phenyl group is sp2 hybridised and there is a partial double bond character. Therefore the attack by I– ion breaks O–CH3 bond to form CH3I. Phenols do not react further to give halides because the sp2hybridised carbon of phenol cannot undergo nucleophilic substitution reaction needed for conversion to the halide.
:
D
In case of anisole, methylphenyl oxonium ion, is formed by protonation of ether. The bond between O–CH3 is weaker than the bond betweenO–C6H5 because the carbon of phenyl group is sp2 hybridised and there is a partial double bond character. Therefore the attack by I– ion breaks O–CH3 bond to form CH3I. Phenols do not react further to give halides because the sp2hybridised carbon of phenol cannot undergo nucleophilic substitution reaction needed for conversion to the halide.
Question 3. Read the assertion and reason carefully to mark the correct option out of the options given below:
(a) If both assertion and reason are true and the reason is the correct explanation of the assertion.
(b) If both assertion and reason are true but reason is not the correct explanation of the assertion.
(c) If assertion is true but reason is false.
(d) If assertion is false but reason is true.
Assertion: The major products formed by heating C6H5CH2OCH3 with HI are C6H5CH, I and CH3OH.
Reason : Benzyl cation is more stable than methyl cation.
(a) If both assertion and reason are true and the reason is the correct explanation of the assertion.
(b) If both assertion and reason are true but reason is not the correct explanation of the assertion.
(c) If assertion is true but reason is false.
(d) If assertion is false but reason is true.
Assertion: The major products formed by heating C6H5CH2OCH3 with HI are C6H5CH, I and CH3OH.
Reason : Benzyl cation is more stable than methyl cation.
Answer: Option A. -> A
:
A
This can be explained on the basis of SN1 mechanism. The carbonium ion produced being benzylium ion and it is more stable than alkylium ion due to resonance.
:
A
This can be explained on the basis of SN1 mechanism. The carbonium ion produced being benzylium ion and it is more stable than alkylium ion due to resonance.
Answer: Option B. -> Conc. H3PO4
:
B
C2H5OH(ethanol) is a very weak acid hence it does not react with NaOH. However, it reacts with metallic sodium, giving sodium ethoxide and hydrogen gas.
:
B
C2H5OH(ethanol) is a very weak acid hence it does not react with NaOH. However, it reacts with metallic sodium, giving sodium ethoxide and hydrogen gas.
Answer: Option B. -> o-and p-bromophenol
:
B
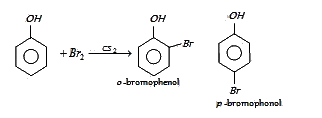
In presence of non-polar solvent (CS2) the ionization of phenol is suppressed. The ring is slightly activated and hence mono substitution occurs with p-bromophenol as the major product.
On the other hand with water phenol forms 2, 4, 6-tribromophenol. (CS2)
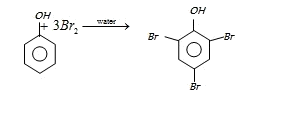
In aqueous solution phenol ionizes to give phenoxide ion. Due to the presence of negative charge of
oxygen the benzene ring is highly activated and hence trisubstituted product is obtained.
:
B
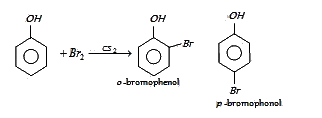
In presence of non-polar solvent (CS2) the ionization of phenol is suppressed. The ring is slightly activated and hence mono substitution occurs with p-bromophenol as the major product.
On the other hand with water phenol forms 2, 4, 6-tribromophenol. (CS2)
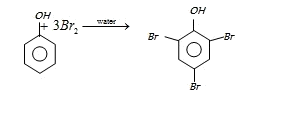
In aqueous solution phenol ionizes to give phenoxide ion. Due to the presence of negative charge of
oxygen the benzene ring is highly activated and hence trisubstituted product is obtained.
Answer: Option C. -> B2H6 followed by H2O2
:
C
When propene reacts with mercuric acetate followed by NaBH4, it gives secondary or tertiary alcohols, in accordance with Markownikoff’s rule. But, with diborane followed by H2O2, it gives primary alcohols which is in accordance with Anti-Markownikoff’s rule. 6CH3−CH=CH2+B2H6H2O2−−−→CH3−CH2−CH2OH
:
C
When propene reacts with mercuric acetate followed by NaBH4, it gives secondary or tertiary alcohols, in accordance with Markownikoff’s rule. But, with diborane followed by H2O2, it gives primary alcohols which is in accordance with Anti-Markownikoff’s rule. 6CH3−CH=CH2+B2H6H2O2−−−→CH3−CH2−CH2OH
Answer: Option D. -> 2-methylpropan-2-ol
:
D
Tertiary alcohols react fastest with hydrogen halides. 2-methyl propan-2-ol is a tertiary alcohol.
:
D
Tertiary alcohols react fastest with hydrogen halides. 2-methyl propan-2-ol is a tertiary alcohol.
Answer: Option C. -> 4-Methoxy-acetophenone
:
C
Friedel-Craft's acylation of anisole with acetyl chloride in presence of anhydrous AlCl3 gives 4-methoxy acetophenone(para) as a major product. Methoxy group in anisole is activating and ortho-para directing with major product being para isomer.
:
C
Friedel-Craft's acylation of anisole with acetyl chloride in presence of anhydrous AlCl3 gives 4-methoxy acetophenone(para) as a major product. Methoxy group in anisole is activating and ortho-para directing with major product being para isomer.
Answer: Option C. -> Chromic anhydride in glacial acetic acid
:
C
Strong oxidising agents like acidified KMnO4 or K2Cr2O7 oxidised alcohols to carboxylic acids. For oxidation of primary alcohols to ketones, Pyridinium chloro-chromate(PCC) is used and for oxidation of secondary alcohols to ketones, Chromic anhydride in glacial acetic acid is used.
:
C
Strong oxidising agents like acidified KMnO4 or K2Cr2O7 oxidised alcohols to carboxylic acids. For oxidation of primary alcohols to ketones, Pyridinium chloro-chromate(PCC) is used and for oxidation of secondary alcohols to ketones, Chromic anhydride in glacial acetic acid is used.