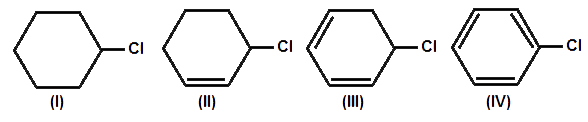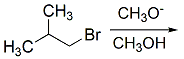11th And 12th > Chemistry
REACTION MECHANISM MCQs
Total Questions : 15
| Page 1 of 2 pages
Answer: Option D. ->
Due to +E effect of Cl
:
D
:
D
Cl is involved in resonance as well as inductive effect and not in electrometric effect as it is more electronegative.
Answer: Option C. ->
II > III > I
:
C
The reactivity decreases in the order
:
C
The reactivity decreases in the order
Allylic > Secondary > primary.
Answer: Option C. ->

:
C

:
C
In protic solvent E2 proceeds over SN2.
Answer: Option D. ->

:
D

:
D
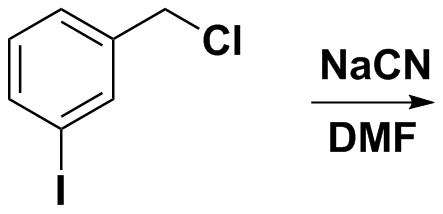
Alkyl halides are more reactive than aryl halides, therefore the halogen present on the side chain is displaced.
Answer: Option D. ->
CH3O−>OH−>CH3COO−>H2O
:
D
:
D
1. Due to the presence of -ve charge CH3O−, OH−,CH3COO− , are more nucleophilic than H2O. Further due to + I effect of the CH3 group, CH3O− is more nucleophilic than OH−.
2. CH3COO− has the -ve charge on oxygen stabilized by resonance and therefore its nucloephilicity is less than those of CH3O− and OH−.
Answer: Option C. ->
DMSO
:
C
:
C
Polar aprotic solvents such as DMSO increase the rate of the SN2 reaction.