12th Grade > Chemistry
NOMENCLATURE OF ORGANIC COMPOUNDS MCQs
Total Questions : 15
| Page 2 of 2 pages
Answer: Option C. -> 2,2-Dimethylpropane
:
C
C5H12 is of the form CnH2n+2.
Hence it is a saturated alkane. Further, it is given that it has a quaternary carbon. Hence its structure is:
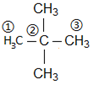
Longest chain = 3 carbons ⇒ word root = prop
Primary suffix = ane
Secondary prefix = 2,2-Dimethyl
IUPAC name is (c) 2,2-Dimethyl propane
:
C
C5H12 is of the form CnH2n+2.
Hence it is a saturated alkane. Further, it is given that it has a quaternary carbon. Hence its structure is:

Longest chain = 3 carbons ⇒ word root = prop
Primary suffix = ane
Secondary prefix = 2,2-Dimethyl
IUPAC name is (c) 2,2-Dimethyl propane
Answer: Option A. -> 3,6-Dimethyl-3-octene
:
A
Parent chain is
1CH3−2CH2−3C|CH3=4CH−5CH2−CH36|CH−7CH2−8CH3
Word root⇒ oct
Double bond⇒suffix⇒ene
Two methyl groups at C3andC5⇒3,6-dimethyl
Therefore, 3,6-Dimethyl-3-octene
:
A
Parent chain is
1CH3−2CH2−3C|CH3=4CH−5CH2−CH36|CH−7CH2−8CH3
Word root⇒ oct
Double bond⇒suffix⇒ene
Two methyl groups at C3andC5⇒3,6-dimethyl
Therefore, 3,6-Dimethyl-3-octene
Answer: Option A. -> 2,4-Dimethylheptane
:
A
Our first step should be to choose the longest chain from the given alkane.
Although there are 9 carbonatoms in total, the longest chain contains only 7 carbon atoms.
Primary suffix =ane
Word root = hept
If we number the chain, as shown above, the two methyl substituents are at C4 and C6
Now, let's look at another case.
Now we have substituents at C2andC4. In both cases, there are exactly two substituents.
We choose thenumbering direction that gives the lowest set of locants.
Hence, secondary prefix = 2,4-dimethyl (since there are two methyl groups-one each at C2andC4
Hence, the IUPAC name is 2,4-Dimethylheptane
:
A
Our first step should be to choose the longest chain from the given alkane.
Although there are 9 carbonatoms in total, the longest chain contains only 7 carbon atoms.

Primary suffix =ane
Word root = hept
If we number the chain, as shown above, the two methyl substituents are at C4 and C6
Now, let's look at another case.

Now we have substituents at C2andC4. In both cases, there are exactly two substituents.
We choose thenumbering direction that gives the lowest set of locants.
Hence, secondary prefix = 2,4-dimethyl (since there are two methyl groups-one each at C2andC4
Hence, the IUPAC name is 2,4-Dimethylheptane