Question
Which of the following are true for modern periodic table?
Answer: Option A
:
A, B, C, and D
In the modern periodic table given below, we can see that all the elements are arranged in the increasing order of their atomic numbers.
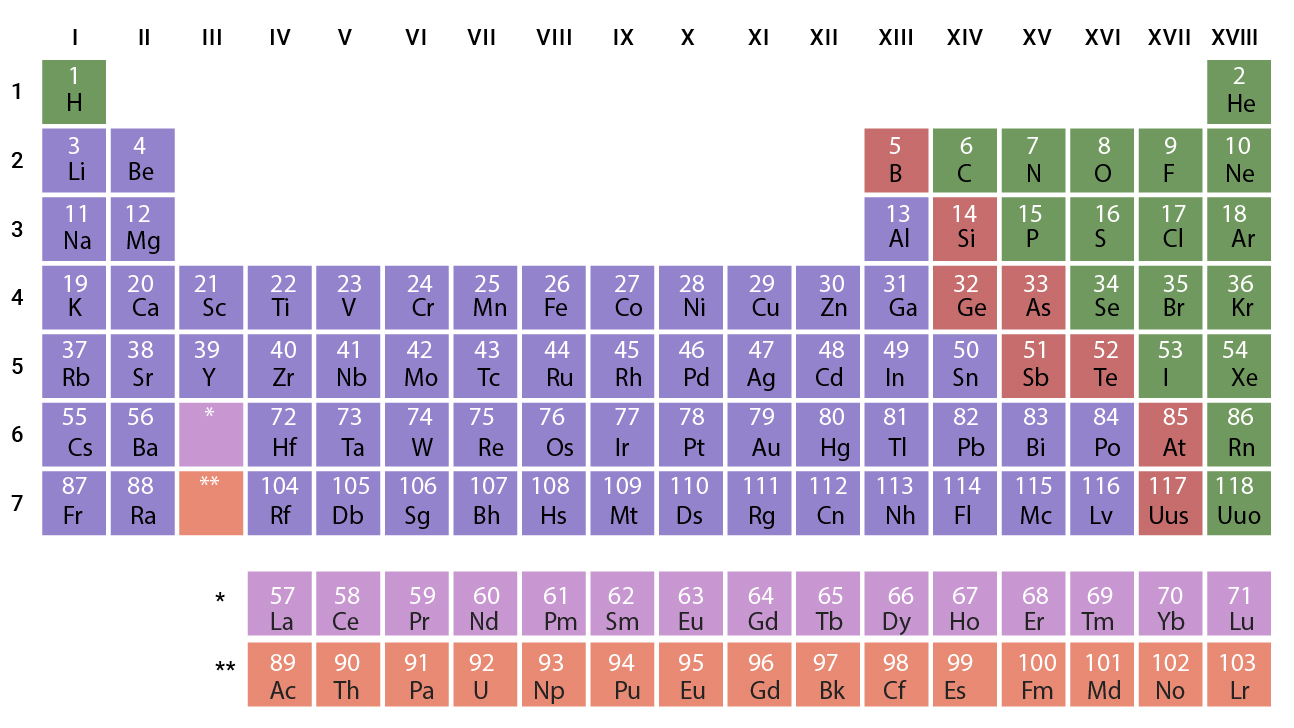
Carbon, for an example, has many isotopes and allotropes. But even so, there is only one place for carbon. Thus, the remaining statements are also true. Also, metals are placed on the left side, non-metals on the right side, and metalloids in between them in a zig-zag manner.
Was this answer helpful ?
:
A, B, C, and D
In the modern periodic table given below, we can see that all the elements are arranged in the increasing order of their atomic numbers.

Carbon, for an example, has many isotopes and allotropes. But even so, there is only one place for carbon. Thus, the remaining statements are also true. Also, metals are placed on the left side, non-metals on the right side, and metalloids in between them in a zig-zag manner.
Was this answer helpful ?


















Submit Solution