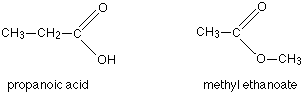12th Grade > Chemistry
ISOMERISM MCQs
Total Questions : 30
| Page 3 of 3 pages
Answer: Option A. -> Metamerism
:
A
CH3COOC2H5andC2H5COOCH3 , the alkyl groups on either side are different. Hence, they are metamers.
The difference is that in the first one a methyl group is attached to the carbon whereas in the second an ethyl group is attached to the carbon.
:
A
CH3COOC2H5andC2H5COOCH3 , the alkyl groups on either side are different. Hence, they are metamers.
The difference is that in the first one a methyl group is attached to the carbon whereas in the second an ethyl group is attached to the carbon.
Answer: Option C. -> 2-Butanol
:
C
Aldehydes and ketones can be functional isomers. But alcohols are not isomeric with aldehydes.
You can just write down the structures of each and count the number of carbons, hydrogens, and oxygens to be certain.
:
C
Aldehydes and ketones can be functional isomers. But alcohols are not isomeric with aldehydes.
You can just write down the structures of each and count the number of carbons, hydrogens, and oxygens to be certain.
Answer: Option B. -> CH3CH=CHCH2CH2CHO
:
B
Compounds A, C and D have same molecular formula as that of CH3−CO−CH2CH2CH2CH3 with a formula of C6H12O
But compound B has C6H10O and hence it is not an isomer.
:
B
Compounds A, C and D have same molecular formula as that of CH3−CO−CH2CH2CH2CH3 with a formula of C6H12O
But compound B has C6H10O and hence it is not an isomer.
Answer: Option D. -> Butanone
:
D
Ketone cannot be an isomer of ether. No matter how you arrange diethyl ether, you cannot get a ketone compound.
First one is simple right? Just remove one carbon on one side and add to the other side.
C2H5−O−C2H5 → C3H7−O−CH3
It can also become an alcohol. We need to add one hydrogen to the oxygen and shift it to the end.
C2H5−O−C2H5 → CH3−CH2−CH2−OH
For third one, follow similar procedure as above. Just use a 3 carbon chain.
:
D
Ketone cannot be an isomer of ether. No matter how you arrange diethyl ether, you cannot get a ketone compound.
First one is simple right? Just remove one carbon on one side and add to the other side.
C2H5−O−C2H5 → C3H7−O−CH3
It can also become an alcohol. We need to add one hydrogen to the oxygen and shift it to the end.
C2H5−O−C2H5 → CH3−CH2−CH2−OH
For third one, follow similar procedure as above. Just use a 3 carbon chain.
Answer: Option B. -> Functional isomerism
:
B
Alcohols and ethers are functional isomers.
:
B
Alcohols and ethers are functional isomers.