12th Grade > Chemistry
HYDROCARBONS MCQs
Total Questions : 28
| Page 3 of 3 pages
Answer: Option D. -> 1,2 -Benzanthracene
:
D
You should be able to see that the 3 benzene rings at the bottom form an anthracene.
And using the lowest locant rule, we can assign 1, 2 to the 2 carbons where benzene is attached.
So, the name would be 1,2-benzanthracene
:
D
You should be able to see that the 3 benzene rings at the bottom form an anthracene.
And using the lowest locant rule, we can assign 1, 2 to the 2 carbons where benzene is attached.
So, the name would be 1,2-benzanthracene
Answer: Option D. -> 1,2 -Benzanthracene
:
A
The compound without branches will havethe maximum surface area and with the increase in surface area, the boiling point also increases.
The answer should be the linear molecule, n-hexane.
:
A
The compound without branches will havethe maximum surface area and with the increase in surface area, the boiling point also increases.
The answer should be the linear molecule, n-hexane.
Answer: Option D. -> 1,2 -Benzanthracene
:
A
addition of water followed by tautomerism
:
A
addition of water followed by tautomerism
Answer: Option C. -> polyacetylene
:
C
A solid-state lithium/iodinebatteryhas been formed by directly contacting metallic lithium with iodine-dopedpolyacetylene
:
C
A solid-state lithium/iodinebatteryhas been formed by directly contacting metallic lithium with iodine-dopedpolyacetylene
Answer: Option A. -> A, B, C only
:
A
A does not undergo any change. There is no strong reason to support KMnO4 attacks a tertiary alkane to give an alcohol
B forms a tertiary alkane through isomerisation
C forms a benzene ring through aromatization reaction
:
A
A does not undergo any change. There is no strong reason to support KMnO4 attacks a tertiary alkane to give an alcohol
B forms a tertiary alkane through isomerisation
C forms a benzene ring through aromatization reaction
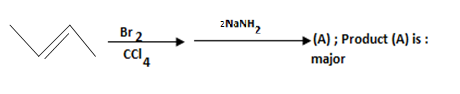
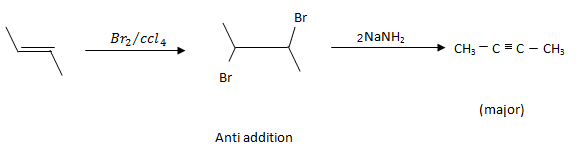
Answer: Option B. -> Pent - 2 - yne
:
B
X is CH3−C≡C−Na+
Y isCH3−C≡C−CH2−CH3
The first step removes the terminal hydrogen from the given substrate (reactant on the left). Such a removable hydrogen is called an acidic hydrogen. In the next step, the nucleophile - which is rich in electrons - attacks the C−Br bond of ethyl bromide. Because of the difference in electronegativity between carbon andbromine, this C−Br bond is polar - meaning, the bonding electrons are closer to the more electronegative bromine. This gives rise to the possibility of the carbon of the C−Br bond having a slight deficit of electrons; let's just say that this carbon misses electrons and that is where the electron-rich terminal carbon of CH3−C≡C− comes in. Hence, the reaction.
:
B
X is CH3−C≡C−Na+
Y isCH3−C≡C−CH2−CH3
The first step removes the terminal hydrogen from the given substrate (reactant on the left). Such a removable hydrogen is called an acidic hydrogen. In the next step, the nucleophile - which is rich in electrons - attacks the C−Br bond of ethyl bromide. Because of the difference in electronegativity between carbon andbromine, this C−Br bond is polar - meaning, the bonding electrons are closer to the more electronegative bromine. This gives rise to the possibility of the carbon of the C−Br bond having a slight deficit of electrons; let's just say that this carbon misses electrons and that is where the electron-rich terminal carbon of CH3−C≡C− comes in. Hence, the reaction.