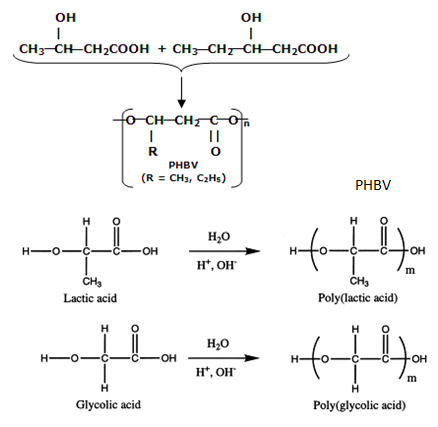
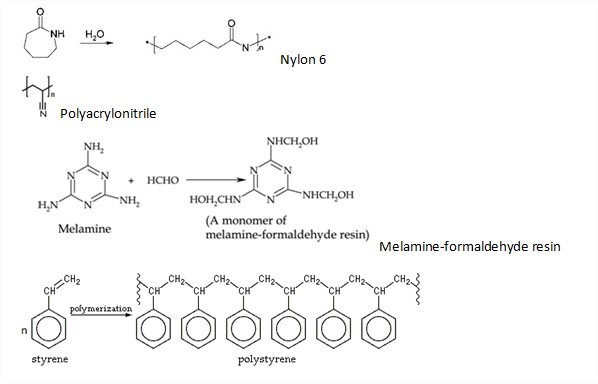
12th Grade > Chemistry
POLYMERS MCQs
Total Questions : 30
| Page 2 of 3 pages
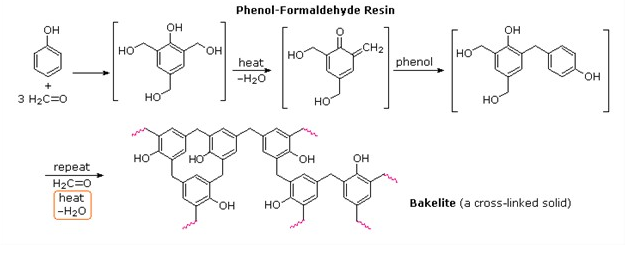
Answer: Option C. -> Bakelite
:
C
Bakelite is thermosetting polymer. It becomes infusible on heating and cannot be remoulded.
:
C
Bakelite is thermosetting polymer. It becomes infusible on heating and cannot be remoulded.
Answer: Option D. -> Both as addition and condensation polymers
:
D
Both as addition and condensation polymers.
:
D
Both as addition and condensation polymers.
Answer: Option B. -> If both assertion and reason are true but reason is not the correct explanation of the assertion.
:
B
Due to the presence of strongC–Fbondsteflon has high thermal stability and chemical inertness.
And it is true that it is a thermoplastic. It changes shape on heating and retains the shape. It cannot be reshaped on further heating.
So, it is true that it is chemically inert but not because it is a thermoplastic.
:
B
Due to the presence of strongC–Fbondsteflon has high thermal stability and chemical inertness.
And it is true that it is a thermoplastic. It changes shape on heating and retains the shape. It cannot be reshaped on further heating.
So, it is true that it is chemically inert but not because it is a thermoplastic.
Answer: Option B. -> Polythene
:
B
Thermoplastic polymers possess intermolecular forces of attraction between elastomers and fibres.Polythene is a thermoplastic polymer.
:
B
Thermoplastic polymers possess intermolecular forces of attraction between elastomers and fibres.Polythene is a thermoplastic polymer.
Answer: Option D. -> Nylon
:
D
Nylon is a synthetic polymer
:
D
Nylon is a synthetic polymer
Answer: Option C. -> Nylon-6,6
:
C
Nylons are types of polyamides.
Even if you didn't know that, if you remember the monomers of nylon-6,6 which are hexamethylene diamine and adipic acid; and if you know about condensation polymerisation, you can figure out the structure of nylon-6,6
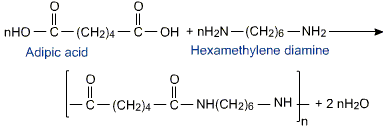
If you remember, an amide is CONH2 ... here we have CONH, this is the amide linkage.
:
C
Nylons are types of polyamides.
Even if you didn't know that, if you remember the monomers of nylon-6,6 which are hexamethylene diamine and adipic acid; and if you know about condensation polymerisation, you can figure out the structure of nylon-6,6
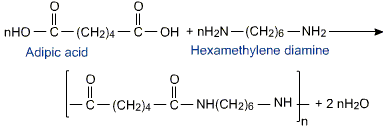
If you remember, an amide is CONH2 ... here we have CONH, this is the amide linkage.
Answer: Option A. -> (−CF2−CF2−)n
:
A
Teflon (−CF2−CF2−)n is stable upto 598K.
:
A
Teflon (−CF2−CF2−)n is stable upto 598K.