Question
Take a thin metallic can half filled with water and put it over the flame so that the water inside starts boiling. Now put the lid on the can and remove it from the flame. What happens to the can when cold water is poured on it?
Answer: Option C
:
C
As soon as the water inside the can is heated, water starts converting to water vapour. On sealing the can, the water vapour inside cannot go out and pressure inside the can increases.
When cold water is poured on the can, the water vapour inside the can condenses into water. Thus, there is a drop in air pressure inside the can. The atmospheric pressure then pushes the can inwards, causing it to distort. This can be visualised from the diagram given below.
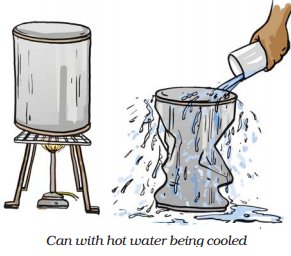
Was this answer helpful ?
:
C
As soon as the water inside the can is heated, water starts converting to water vapour. On sealing the can, the water vapour inside cannot go out and pressure inside the can increases.
When cold water is poured on the can, the water vapour inside the can condenses into water. Thus, there is a drop in air pressure inside the can. The atmospheric pressure then pushes the can inwards, causing it to distort. This can be visualised from the diagram given below.

Was this answer helpful ?


















Submit Solution