Question
Answer: Option B
:
B
Decide the parent chain and give the numbering so that more substituted substituent will get the least number.
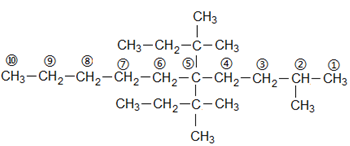
parent chain
10 carbons ⇒ word root ⇒ dec
single bond ⇒ suffix ⇒ ane
substituents at C5andC2
Now,
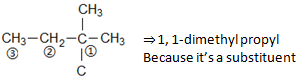
There is one more same substituent on C5
Therefore,
5,5-Bis-(1,1-dimethyl propyl)
Bis⇒ Because two same substituents are there, and a methyl group at C2⇒ 2-methyl
Therefore,
5,5-Bis-(1,1-dimethylpropyl)-2-methyldecane
Was this answer helpful ?
:
B
Decide the parent chain and give the numbering so that more substituted substituent will get the least number.

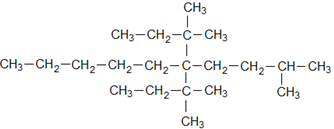
parent chain
10 carbons ⇒ word root ⇒ dec
single bond ⇒ suffix ⇒ ane
substituents at C5andC2
Now,
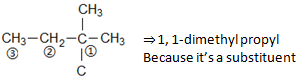
There is one more same substituent on C5
Therefore,
5,5-Bis-(1,1-dimethyl propyl)
Bis⇒ Because two same substituents are there, and a methyl group at C2⇒ 2-methyl
Therefore,
5,5-Bis-(1,1-dimethylpropyl)-2-methyldecane
Was this answer helpful ?
More Questions on This Topic :
Question 3. The IUPAC name of the following compound is....
Question 4. The IUPAC name of the following compound is?....
Question 7. The IUPAC name of....
Question 8. The IUPAC name of....
Question 9. The IUPAC name of this compound is? ....


















Submit Solution